Research
Design and synthesis of organosilicon compounds with novel structures
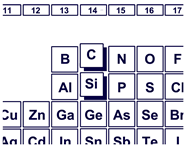 Properties of carbon (the simbol C) are basically similar to those of silicon (the simbol Si), because carbon and silicon both belong to Group 14 in the periodic table. However, the chemistry of silicon differs from that of carbon, although carbon and silicon both have a normal valence of four. Our interest lies in the design and synthesis of organic molecules with new structures that incorporate silicon to develop molecules with new functions. Specifically, we are studying the relationship between molecular structure and molecular functionalities of novel organosilicon compounds.
Properties of carbon (the simbol C) are basically similar to those of silicon (the simbol Si), because carbon and silicon both belong to Group 14 in the periodic table. However, the chemistry of silicon differs from that of carbon, although carbon and silicon both have a normal valence of four. Our interest lies in the design and synthesis of organic molecules with new structures that incorporate silicon to develop molecules with new functions. Specifically, we are studying the relationship between molecular structure and molecular functionalities of novel organosilicon compounds.
Molecular Gyrotop
Macrocage molecules with a bridged rotor have been synthesized as molecular gyroscopes and molecular gyrotops given that the rotor can rotate even in the crystalline state.
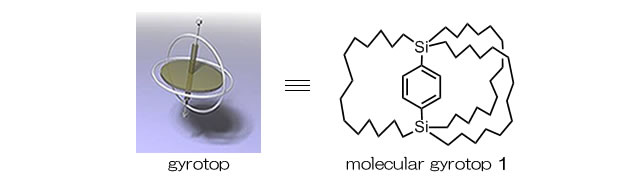
Modulation of the Birefringence: We reported thermal modulation of the birefringence of a molecular gyrotop 1 crystal possessing a phenylene rotor encased in three long alkyl-spokes as the first application of variation in the optical properties because of the dynamics of the phenylene rotor in a crystal.
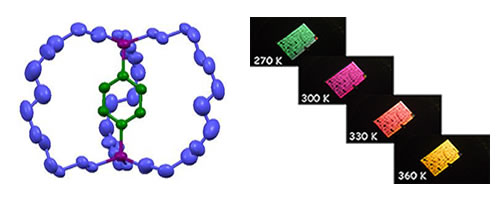
W. Setaka, K. Yamaguchi, PNAS, 109, 9271-9275 (2012). doi:10.1073/pnas.1114733109 (Highlighted in ASAHI newspaper.) (Press Release : Tokushima Bunri University)
Molecular Balloon : Although the most stable cage structure of the molecular gyrotop 2 in a crystal was folded and shrunken at low temperature, expansion of the cage was observed at high temperature due to rapid rotation of the phenylene in a crystal. This phenomenon is analogous to the deflation and inflation of a balloon.
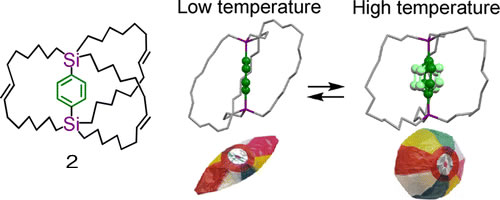
W. Setaka, K. Yamaguchi, JACS, 134, 12458-12461 (2012). doi:10.1021/ja305822e (Highlighted in Nature Chemistry)
Order-Disorder Transition of Dipolar Rotor in the Crystalline State: A thiophene-bridged macrocage molecule 3 was synthesized as a molecular gyrotop having a dipolar rotor. The thermal change in the orientation of the dipolar rotors (thiophene ring) inside the crystal, i.e., order–disorder transition, was observed.
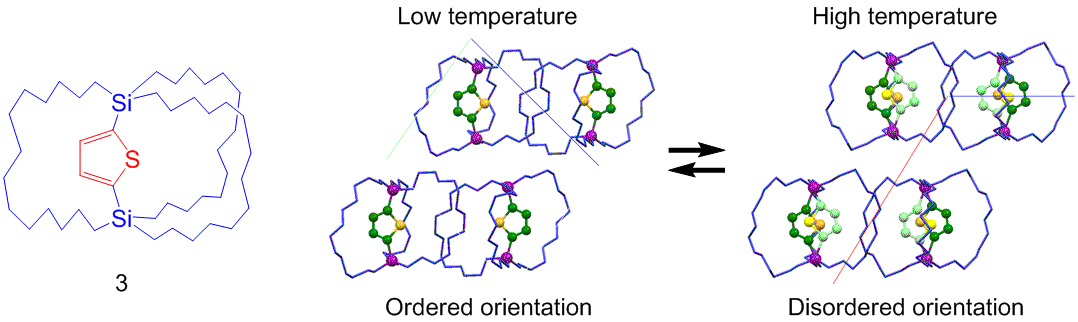
文献: W. Setaka, K. Yamaguchi, JACS, 135, 14560-14563 (2013). doi:10.1021/ja408405f
Molecular Shuttle
We reported a silicon-based directed synthesis of [2]rotaxanes composed of carbon, hydrogen, and silicon atoms, and hence, without polar functional groups. Facile shuttling of the wheel was observed to control the axle conformation in solution.
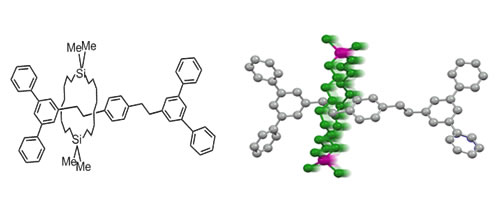
S. T. Phan, W. Setaka, M. Kira, CL, 37, 976-977 (2008). doi:10.1246/cl.2008.976
Molecular Gear
Since pioneering studies of Mislow et al. and Iwamura et al., bis(9-triptycyl)X systems (X = CH2, O, NH, SiH2, PH, etc.) are known as well-designed bevel gear systems. We have reported that the introduction of the clutch-declutch mechanism into a new gear system, a bis(9-triptycyl)difluorosilane derivative, is achieved by the reversible attachment of a fluoride ion and that the corresponding pentacoordinate silicate adopts an unusual trigonal bipyramid (TBP) structure having both the two 9-triptycyl groups at the apical positions against the Muetterties rule.
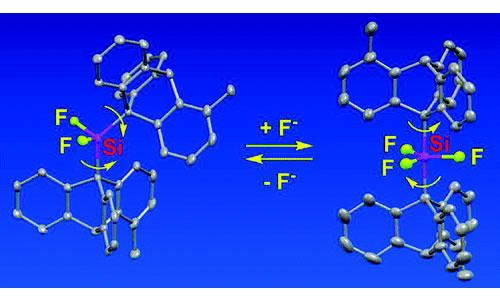
W. Setaka, T. Nirengi, C. Kabuto, and M. Kira, JACS, 130, 15762-15763 (2008). doi:10.1021/ja805777p